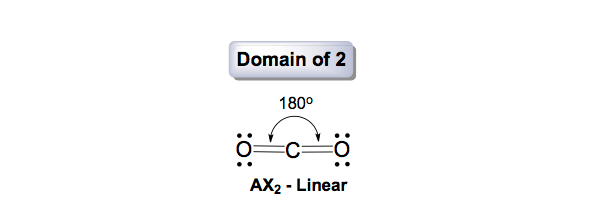
Therefore, the ideal geometry should be tetrahedral (109.5⁰). Since the molecule NCl 3 contains 3 bond pairs and one lone pair, its geometry will be trigonal pyramidal. These two electrons will form one lone pair of electrons.ĥ. Since 3 electrons of nitrogen are used to form single covalent bonds, 2 electrons remained of the total 5. 2 electrons are needed to form a single lone pair. The remaining electrons form lone pair(s), if any. Moreover, once all the σ-bonds have been assigned, the π-bonds should be considered depending on the valency of the bonded atoms.Ĥ. In the example provided, since 3 chlorine atoms are bonded with the central nitrogen atom, 3 electrons are required of the 5 valence electrons of N, to form 3 bond pairs. One electrons is required from the central atom to form a bond pair. The electrons are allotted to the number of bonds formed. Nitrogen is a group 5A element, and so possesses 5 valence electrons.Īdditionally, considering the formal charge on the central atom, an electron is added for each negative charge, and an electron is subtracted for each positive charge, if applicable.ģ. Once the central atom has been identified, the number of electrons present in its valence shell is considered. For example, in the case of NCl 3, nitrogen is the central atom.Ģ. It is usually the atom that occurs once in the molecular formula. The first step in determining the shape of molecules is to identify the central atom from the molecule. At the vertices of the base lie the 3 hydrogen atoms and the top of the pyramid is the nitrogen atom with its lone pair.ĭetermining Molecular Geometries and Bond Angles by VSEPR Theoryġ. The shape is a pyramid with a triangular base. For example, the NH 3 molecule assumes a trigonal pyramidal geometry. The geometries are exactly what they sound like. Some common VSEPR shapes are: linear, trigonal planar, bent, tetrahedral, trigonal pyramidal, trigonal bipyramidal, and octahedral. The VSEPR theory helps us to predict structures of various molecules. The detailed postulates of VSEPR theory are provided at the end of the article. Shapes of molecules depend upon the number of bond pairs and lone pairs, and this information is provided in a VSEPR table/chart. Double and Triple bonds occupy more space, and cause more repulsion but are considered as single bonds in determining the geometry.Ħ. Both Lone pairs and Bond pairs contribute in determining the geometry.ĥ. 
The order of repulsion between the electron pairs is: L.P – L.P > L.P – B.P > B.P – B.P.Ĥ. sy2), Tripos Sybyl Line Notation (.sln), Beilstein ROSDAL (.ros), XYZ Files (.3. 
mmod), Schrödinger Maestro (.mae), Standard Molecular Data (.smd), Tripos Mol2 (.mol2. ent), RCSB Protein Data Bank Markup Language (.xml. mmcif), RCSB MacroMolecular Transmission Format (.mmtf), RCSB Protein Data Bank Files (.pdb. rd), MDL RXNFiles, both V2000 and V3000 connection tables (.rxn), MMI SketchEl Molecule (.el), Molinspiration JME String (.jme), RCSB Binar圜IF (.bcif), RCSB Macromolecular Crystallographic Information File (.cif. dx), ISIS Sketch File (.skc), ISIS Sketch Transportable Graphics File (.tgf), MDL MOLFiles, both V2000 and V3000 connection tables (.mol. com), IUPAC InChI (.inchi), IUPAC JCAMP-DX (.jdx. 
Read and write many popular chemical file types for working with the applications you use:ĪCD/ChemSketch Documents (.sk2), ChemDoodle Documents (.icl), ChemDoodle 3D Scenes (.ic3), ChemDoodle Javascript Data (.cwc.js), CambridgeSoft ChemDraw Exchange (.cdx), CambridgeSoft ChemDraw XML (.cdxml), Crystallographic Information Format (.cif), CHARMM CARD File (.crd), ChemAxon Marvin Document (.mrv), Chemical Markup Language (.cml), Daylight SMILES (.smi. Algorithmic Analysis of Cahn−Ingold−Prelog Rules of Stereochemistry: Proposals for Revised Rules and a Guide for Machine Implementation. and is 100% accurate in all 300 test cases provided. The CIP algorithm in ChemDoodle is validated against the test suite provided by Hanson et. Stereochemical features in your structures will be assigned "R", "S", "E", "Z", "M" and "P" descriptors. to remove any ambiguities and describe a completely consistent system for CIP assignments.ĬhemDoodle implements all 6 current CIP rules as well as auxilliary desciptors and mancude ring support. The most recent CIP rules from IUPAC were then algorithmically analyzed and standarized by Hanson et al. These rules were adopted by IUPAC for naming standards and fully described in the Blue books. While flawed, they have seen many revisions over the decades and were clarified by the work of Paulina Mata. The CIP rules have long been the standard for describing configurations of stereochemical features in a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
